Depression Clinical Trials: What Participants Should Know About Research Studies
Mental health research studies continue across the country, offering participants access to experimental treatments while contributing to depression research. These trials involve various phases of testing, from initial safety studies to large-scale effectiveness research. Participants work with medical teams, follow specific protocols, and help researchers understand how new treatments perform compared to existing options.

Understanding Depression Research Studies
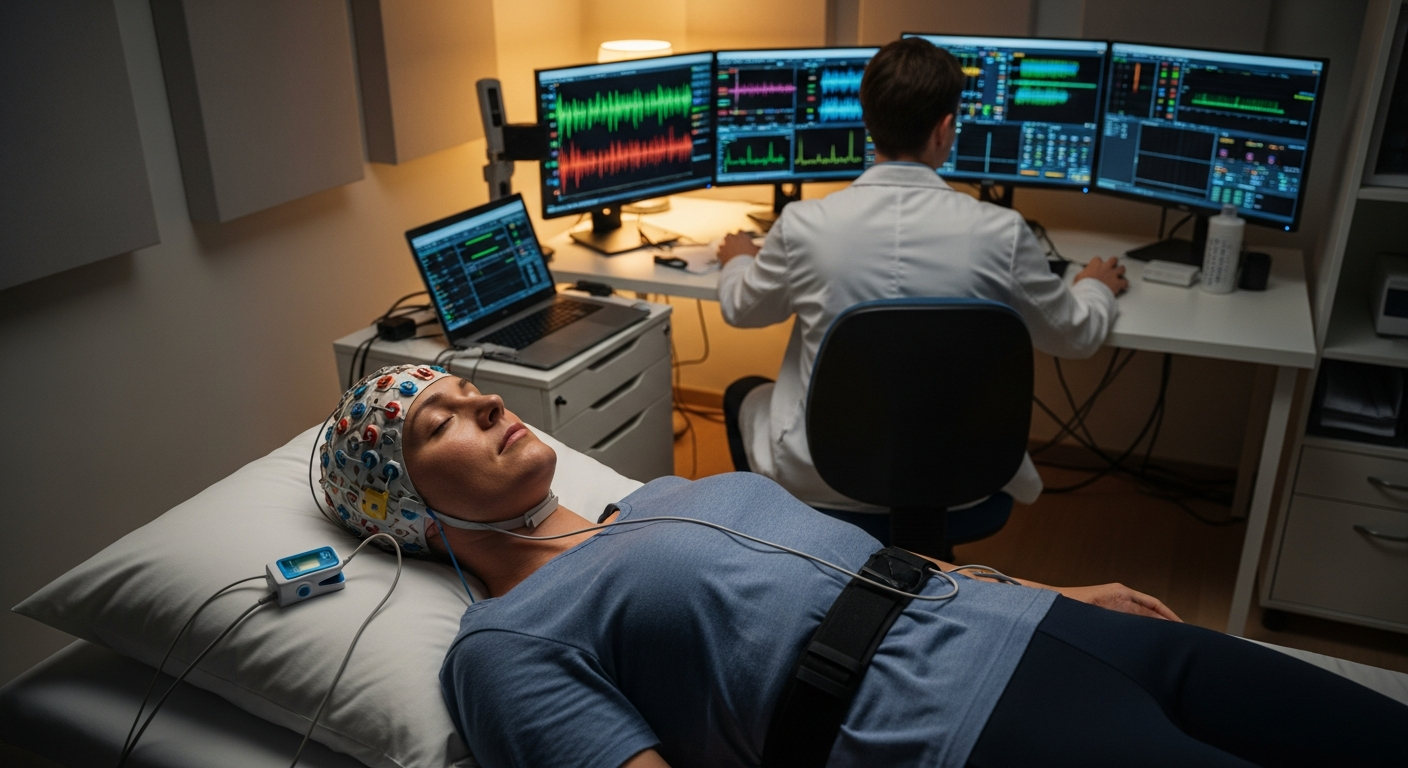
Depression research studies encompass various types of clinical investigations designed to improve our understanding and treatment of depressive disorders. These studies may evaluate new antidepressant medications, compare existing treatments, test psychotherapy approaches, or examine biological markers associated with depression. Researchers conduct these trials in phases, starting with small safety studies and progressing to larger effectiveness trials involving hundreds or thousands of participants. The National Institute of Mental Health and pharmaceutical companies fund most depression clinical trials, which take place at universities, hospitals, and specialized research centers across the country.
Clinical Trial Participation Requirements and Process
Participating in a depression clinical trial typically begins with a screening process to determine eligibility. Researchers establish specific inclusion and exclusion criteria based on factors like depression severity, previous treatments, age, and other health conditions. Potential participants undergo comprehensive evaluations including psychiatric assessments, medical history reviews, and sometimes laboratory tests. The informed consent process ensures individuals understand the study purpose, procedures, potential risks, expected benefits, and their right to withdraw at any time. Most trials require regular visits to the research site for assessments, which may include questionnaires, interviews, and monitoring of symptoms and side effects.
Mental Health Research Protections and Safety Measures
Ethical guidelines and federal regulations protect participants in mental health research. Institutional Review Boards review and approve all clinical trials before they begin, ensuring the study design minimizes risks and maximizes potential benefits. Participants receive detailed information about the study and must provide voluntary informed consent. Researchers monitor participants closely throughout the trial, with protocols in place to address worsening symptoms or adverse events. Studies typically include safety boards that review ongoing data and can halt trials if concerns arise. Participants maintain the right to continue their regular mental health care and can leave a study at any point without affecting their standard treatment options.
Experimental Depression Treatments Being Studied
Current depression clinical trials investigate diverse treatment approaches beyond traditional antidepressants. Researchers are studying novel medications targeting different brain pathways, including fast-acting treatments that may provide relief within hours rather than weeks. Other areas of investigation include neuromodulation techniques like transcranial magnetic stimulation, ketamine-based therapies, psychedelic-assisted treatments, digital therapeutics, and personalized medicine approaches based on genetic profiles. Some trials focus on treatment-resistant depression, examining combination therapies or interventions for individuals who have not responded to standard treatments. Studies also explore prevention strategies, early intervention programs, and treatments tailored to specific populations such as adolescents, older adults, or people with co-occurring medical conditions.
Comparing Research Participation Options
Individuals considering clinical trial participation have various options depending on their location, depression type, and treatment history. Different trials offer distinct approaches and requirements that potential participants should carefully evaluate.
| Study Type | Research Focus | Typical Duration | Key Considerations |
|---|---|---|---|
| Medication Trials | New antidepressants or drug combinations | 8-16 weeks | May include placebo group; regular monitoring required |
| Psychotherapy Studies | Cognitive behavioral therapy or other talk therapy approaches | 12-20 weeks | Multiple weekly sessions; homework assignments common |
| Neuromodulation Research | Brain stimulation techniques like TMS | 4-8 weeks | Daily or frequent visits; non-invasive procedures |
| Observational Studies | Natural course of depression without intervention | 6-24 months | Less intensive; minimal treatment changes |
| Prevention Trials | Interventions for at-risk individuals | Variable | May target specific populations; long-term follow-up |
Prices, rates, or cost estimates mentioned in this article are based on the latest available information but may change over time. Independent research is advised before making financial decisions.
Benefits and Considerations for Trial Participants
Participating in depression clinical trials offers several potential advantages alongside important considerations. Participants often receive close monitoring from experienced mental health professionals, access to new treatments before they become widely available, and comprehensive assessments of their condition at no cost. Many studies provide compensation for time and travel expenses. Contributing to scientific knowledge that may help others with depression can provide a sense of purpose. However, participants should consider potential drawbacks including time commitments for frequent visits, possibility of receiving placebo instead of active treatment, unknown side effects of experimental interventions, and additional paperwork and assessments beyond standard care. The decision to participate should involve careful discussion with healthcare providers, family members, and the research team.
Clinical trials remain essential for advancing depression treatment and improving outcomes for millions of people affected by this condition. Informed participation helps drive medical progress while potentially offering individuals access to innovative therapies and expert care. Anyone considering joining a depression research study should thoroughly review study details, ask questions, and ensure the commitment aligns with their treatment needs and personal circumstances.
This article is for informational purposes only and should not be considered medical advice. Please consult a qualified healthcare professional for personalized guidance and treatment.